Section 1 | An Overview of Necrotic Enteritis
Industry
Page 05 /
Agent Factors
Clostridium perfringens
In order to see necrotic enteritis (NE) in a group of birds, C.perfringens infection needs to be present. There are many strains of C.perfringens, and they vary in their ability to damage the intestine and cause disease in birds1. Practically, what this means is that there are some forms of C.perfringens that allow them to out-compete other “good” bacteria and cause damage to the intestines. The process through which C.perfringens causes disease (otherwise known as its pathogenesis) is summarized in the following figure:
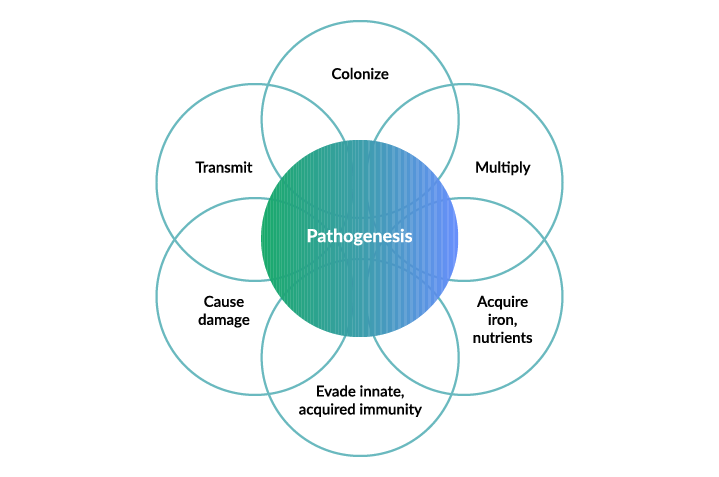
Figure: The disease process of NE in poultry2
Understanding Pathogenesis
Infection with C.perfringens is through the fecal-oral route — birds consume the bacteria from a contaminated environment. C.perfringens then establishes itself (colonizes) and multiplies in the intestines of birds, acquiring iron and other nutrients essential for growth. After this, C.perfringens need to find a way to evade the immune defenses of the bird, allowing them to grow in numbers and damage the intestines. C.perfringens causes this damage, including production of toxic gases in tissue, severe inflammation, platelet clotting (which, in turn, prevents blood clotting, allowing for bleeding to occur), and damage to the cells lining the intestines preventing absorption of nutrients, through the production of necrotic enteritis B-like toxins (NetB)3.
To complete the disease cycle, in infected birds, C.perfringens is shed in the manure, contaminating the environment, allowing it to transmit and infect other birds.

Source: The Poultry Site
Coccidiosis: A gateway to NE
There are a variety of “good” bacteria present in the gastrointestinal tract of birds — they play an important role in digestion, growth, and immune function. Collectively, bacteria, viruses, and protozoa that live in the guts of animals are called the microbiota.
Infection with the parasite Eimeria spp. (also known as coccidia) can play a key role in disrupting the protective gut microbiota, setting the stage for C. perfringens to invade, proliferate, and cause disease1. Coccidiosis also damages the gut leading to the release of a significant amount of protein. If C. perfringens is there, it gives them a valuable source of nutrition and allows their numbers to grow1.
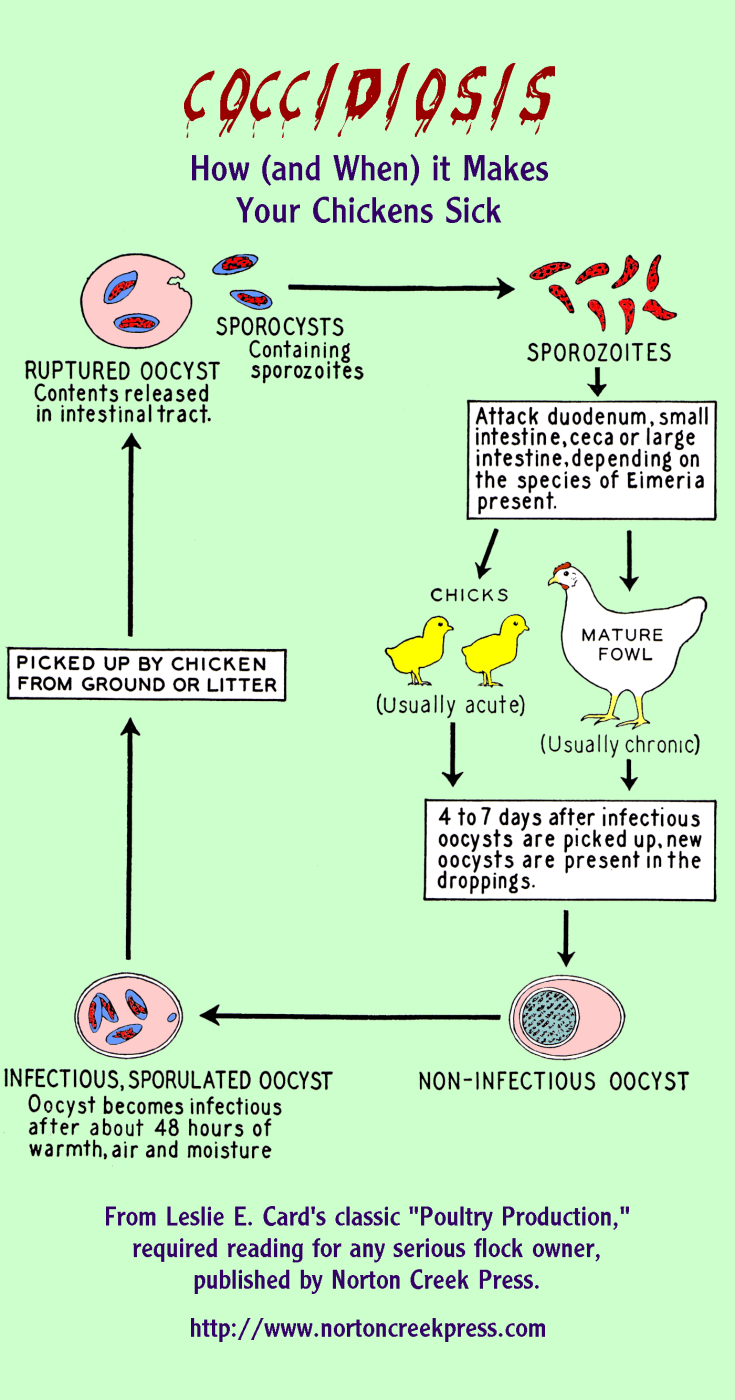
Source: Practical Poultry Tips
Other Bacteria
Infection with other disease-causing bacteria can also predispose birds to NE. For instance, Salmonella infections are found to increase NE severity and death rates1. These effects could be due to intestine damage allowing other bacteria to penetrate the intestinal cells or through suppression of the bird’s immune system1, reducing their capacity to fight off future infection.
Ask the expert:
Canadian poultry industry experts strongly recommend ensuring the water is tested at breeding facilities. Reducing bacterial exposure in water to breeders will reduce the number of egg yolk sac infections that occur, and can help to improve chick health at placement.
Where producers can have an impact is ensuring that water quality is tested, lines are cleaned, pH is appropriate, etc. This must be in place prior to the introduction of a crop of birds. Water quality is KEY to gut health, thereby reducing the risk for necrotic enteritis.
Some water measurement recommendations and targets include:
- pH of 6 or lower
- Free of chlorine
- Add soap and disinfectant between flocks, and ensure water lines are flushed thoroughly
- Quality assurance programs require that nipples be triggered to ensure effective disinfection
- Look in water lines for signs of biofilm accumulation and calcification
For some information on water testing, check out these videos by the Chicken Farmers of Canada:
References
- Moore RJ. Necrotic enteritis predisposing factors in broiler chickens. Avian Pathol. 2016;45(3):275–81.
- Prescott JF, Parreira VR, Mehdizadeh Gohari I, Lepp D, Gong J. The pathogenesis of necrotic enteritis in chickens: what we know and what we need to know: a review. Avian Pathol. 2016;45(3):288–94.
- Abid SA, Azeem T, Chaudhary ZI, Ahmad MUD, Rehman ZU, Umar S. Emerging threat of necrotic enteritis in poultry and its control without use of antibiotics: A review. J. Anim. Plant Sci. 2016;26(6):1556–667.